SEATTLE, Jan. 23, 2020 /PRNewswire/ — Bardy Diagnostics, Inc., (“BardyDx”), a leading provider of ambulatory cardiac monitoring technologies and custom data solutions, announced today the commercial launch of the 14-Day version of the Carnation Ambulatory Monitor (“CAM™”), the industry’s only P-wave centric™ ambulatory cardiac patch monitor and arrhythmia detection device, following recent clearance by the FDA.
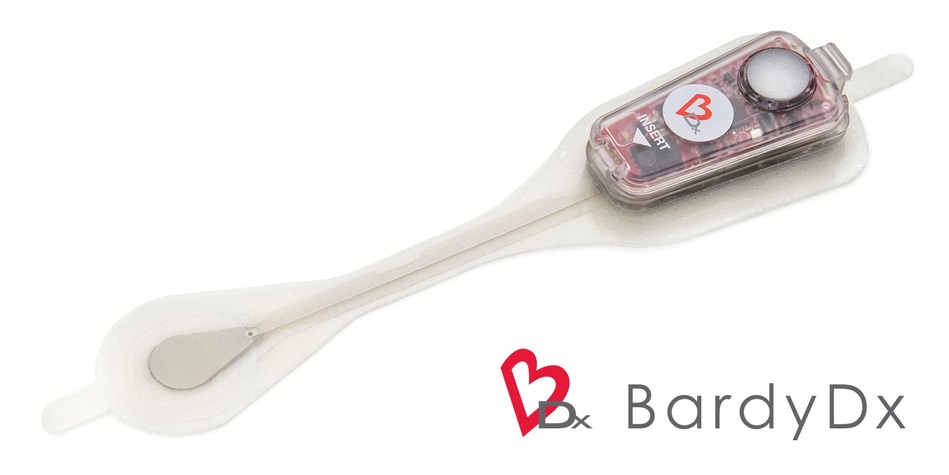
“The 14-Day CAM is the culmination of years of development focused on uncovering the full complexity and meaning of a patient’s cardiac rhythm,” said Gust H. Bardy, MD, Founder and Chief Executive Officer of BardyDx. “We are proud to develop and introduce the most advanced and accurate cardiac monitoring technologies that enable new opportunities to reimagine and redefine patient care.”
The significance of the CAM Patch’s P-wave centric clinical value was highlighted in the American Heart Journal that published the results of a head-to-head comparison with the iRhythm Zio® XT patch, concluding that the BardyDx CAM Patch identified 40% more arrhythmias and resulted in better, more informed clinical decision-making in 41% of patients over the iRhythm Zio XT patch. In addition, a preceding study also published in the American Heart Journalcomparing the CAM Patch and a traditional Holter monitor, showed a four times increase in arrhythmia detection using the CAM Patch, including arrhythmias missed or incorrectly identified using the Holter monitor, concluding that the CAM Patch offered significantly improved rhythm diagnostics.
Strong market uptake of BardyDx’s P-wave centric solutions has spurred recent expansion of the company’s infrastructure to maintain its rapid pace. A second ECG monitoring center located in New Jersey was established last quarter, which received Medicare enrollment approval by the Centers for Medicare & Medicaid Services effective December 2, 2019. In addition, BardyDx recently announced the addition of medical device industry veteran, Ed Vertatschitsch, as Chief Operating Office, along with several new vice president appointments to strengthen the Leadership Team.
“We are extremely proud of the continued momentum of the CAM Patch becoming the cardiac monitor of choice and trusted solution of cardiologists and electrophysiologists across the U.S., U.K., and Canada,” said Ken Nelson, Chief Commercial Officer. “It is increasingly clear that customers are realizing the true clinical and market-differentiating value of our P-wave focused detection technology in enabling optimal patient care.”
The growing market recognition of the innovative P-wave centric CAM Patch includes recently being selected as winner of the Remote Monitoring in Arrhythmias Digital Health Pitch Session at European Society of Cardiology Congress 2019 and finalist of the UCSF Digital Health Award for Best Cardiovascular Digital Diagnostic. In addition, BardyDx was also named the winner of the 2019 MedTech Breakthrough Award for Best New Diagnostic Technology and the winner of the 2019 Frost & Sullivan Award for Technology Innovation in Remote Cardiac Monitoring.
About Bardy Diagnostics:
Bardy Diagnostics, Inc. is an innovator in digital health and remote patient monitoring, with a focus on providing the most diagnostically-accurate and patient-friendly cardiac monitors to the industry. The company’s CAM Patch is a non-invasive, P-wave centric™ ambulatory cardiac monitor and arrhythmia detection device that is designed to improve patient compliance for adults and children through its lifestyle-enabling form factor. Designed to be worn comfortably and discreetly for up to 14 days, the female-friendly, hourglass-shaped CAM Patch is placed on the center of the chest, directly over the heart for optimum ECG signal collection. The proprietary technology of the CAM Patch provides optimal detection and clear recording of the often difficult-to-detect P-wave, the signal of the ECG waveform that is essential for accurate arrhythmia diagnosis. For more information, please visit www.bardydx.com.
Related Links
PRNewswire
